
A Pictorial Guide to the correlation between emotions and cloud formations
Shannon May is an illustrator living in Baltimore, MD. She writes that she is "fascinated by the promise and aesthetics of science and loves exploring, being uncomfortable, books, clouds, and geometry." Her intersection of art and science is up my alley. In fact, her portrait of Heisenberg reminds me of my own approach to portraits of scientists: combining their face with their work. At first I thought this was not that literal, that the dots were merely molecules. But, if you look closely, you can see that she is specifically trying to illustrate the nature of the Heisenberg Uncertainty Principle (no mean feet). One formulation,
Δx Δp ≥ ħ/2
appears at the bottom. This inequality means that the product of the uncertainty in the position of ANYTHING and the uncertainty in its momentum is greater or equal to a half h-bar (Planck's constant divided by 2π, or ħ=h/2π). This means we can never know the position and momentum (mass times velocity, or basically, the motion) of ANYTHING with absolute precision. It turns out that Planck's constant is very small, so this limit on the knowability of both location and motion is largely irrelevant to everyday life of things we can see (people, trees, planets, cars, mice, or even bacteria). But, in the quantum world of the very small, this limit has profound implications. The only way we can 'see' the very small, for instance, an electron (we'll call him Bob), involves hitting Bob with at least a single quantum of light (a photon). But if you hit Bob with a photon he'll go off running in all directions, since the photon will transfer some momentum to Bob - so, we might know exactly where Bob was, when he was hit with the photon, but we don't know his momentum at all. Conversely, we could measure Bob's collision with another particle or photon and know his momentum but we could no longer know where he was. Also we can know both position and momentum, but only with a certain fuzziness or lack of precision (dictated precisely by the inequality above). Thus, we can precisely describe behaviours of groups of small things in a statistical way, but it is inherently impossible to precisely predict the behaviour of individual quanta like Bob.

I think in this illusration, Shannon May is trying to show this with her filled and hollow circles, to denote positions (filled) and posible positions (hollow) of particles and their interactions shown by little red arrows.
Other illustrations like A Pictorial Guide to the correlation between emotions and cloud formations seem both humourous metaphor, with a wink and a tip of the hat to the science of cloud physics. Some are straightforward wonder at astronomy.

illustration for Italo Calvino's 'Cosmicomics'
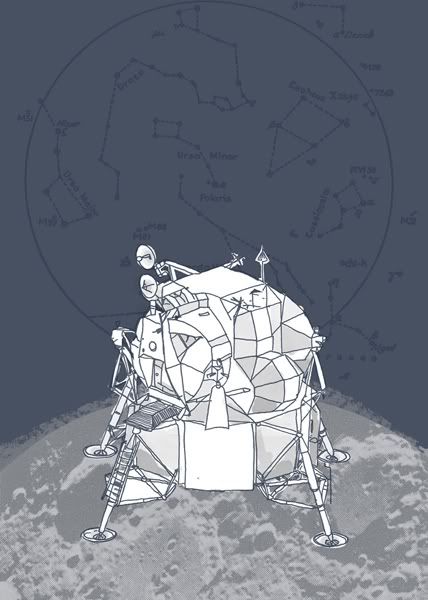
Lunar Park, personal work

Illustration for article Contrarian Investor Sees Economic Crash in China

Music for Airports, illustration for article about the history of rock music
I love how that last one combines the sound waveform and airplane steam trail.
You can find her site, her blog and an etsy shop for craftier endeavours and check out the rest of her work.
{via both design sponge and the shallow end}






This comment has been removed by a blog administrator.
ReplyDelete